 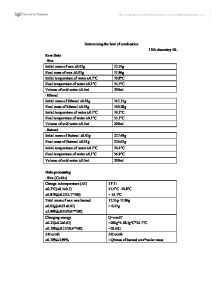
Avoid touching the base of the copper can.Ģ.Leave adequate time for the equipment to cool before touching.Extinguish the flame by replacing the cap of the spirit burner.When not in use, keep the alcohol away from the ignition source.Outline two safety risks in this experiment. Let’s investigate the safety, errors, reliability and accuracy of this experiment.ġ. Step 1: Write a balanced chemical equation of the process. The steps which can be used to determine the enthalpy changes of combustion are outlined below: The standard enthalpy of combustion \small (\Delta H_c^\circ) is the enthalpy change when one mole of a substance undergoes complete combustion with oxygen at standard states, under standard conditions. To determine the enthalpy of combustion of fuels using a calorimeter. This Year 11 Chemistry practical report on the calorimetry experiment consists of: In this article, you will find a complete Chemistry practical report on determining the enthalpy of combustion of fuels via calorimetry. Not only is this experiment commonly performed by students during their Year 11 Chemistry course but also in the HSC Chemistry course. How to perform the calorimetry experiment in Year 11 Chemistry PracticalĪ popular Year 11 Chemistry practical investigation is the calorimetry experiment. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
